Getting ISO 13485 Certified in Woodbridge Township, New Jersey (NJ)
Contact Us

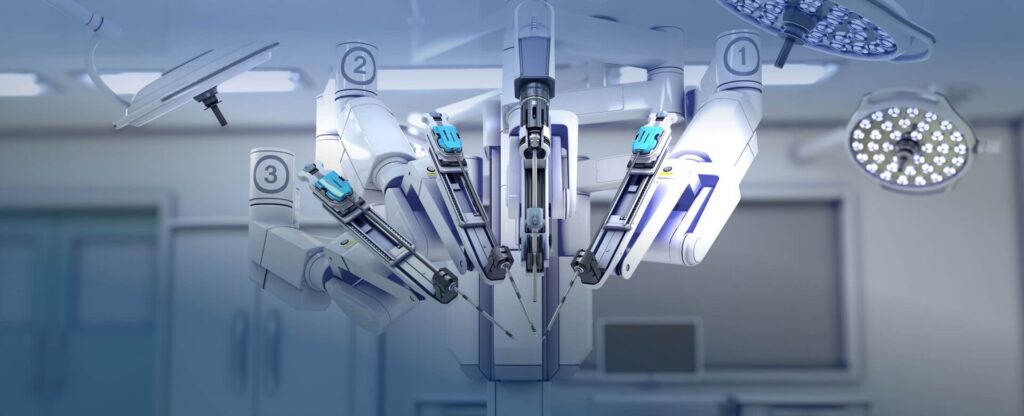
ISO 13485 is a comparable standard to ISO 9001 and specifically acts as a reference for reviewing best practices in the area of safety. This International Standard lays out the quality management system (QMS) standards that can be utilized by an organization to design and build, construct, operate medical devices, design, create and provide similar services. When getting ISO 13485 certified in Woodbridge Township, New Jersey (NJ), our team at IQC ISO Pros will be able to assist you with training and certification for your organization.
The certification provides a particular solution to the development and selling of medical products, and most medical practitioners have selected manufacturers and producers certified by ISO 13485. The standardization framework reflects a worldwide initiative to facilitate the professional manufacture of Medical Devices in standard with world-class management standards which is all in compliance with ISO 13485:2016.
The rationale included in the compliance audit involves the manufacture and selling of medical equipment and awareness of the future damage involved in its application. The certification permits providers of medical equipment to meet with and comply with universal health standards in developed countries. The standard incorporates management frameworks to evaluate medical product production and includes input about how to strengthen the shortcomings of the method.
Criteria of certification ISO 13485:2016 are also extensive, and they aim to increase the development and distribution of medical equipment to a high standard. A control framework under ISO 13485 can be used as a framework to maintain compliance with relevant customer and regulatory criteria.
Phases of Certification
Similar to other ISO certifications, manufacturers of medical equipment requiring clearance in 13485 would first be told of regulatory specifications and their impact on their customers, and of the consequences of a compliance scheme. A management framework that complies with the requirements of the standard must also be implemented inside the organization.
First of all, a quality manual will be developed; the quality manual will define the company’s goals, policies, and procedures for quality control and execution. A person with the know-how to plan and implement such a program may internally construct the system, otherwise, a skilled auditor with expertise in the industry of 13485 may be needed. In Woodbridge Township, New Jersey (NJ), you’ve got IQC The ISO Pros to call on.
The organization will look for an accreditation agency it is satisfied with after writing the qualitative document and implementing a compliance process. When applying to an accrediting authority, the organization must be assured that its accrediting entity has 13485 certificates approved by the Registrar. The organization that certifies would request that its credentials and references be submitted to a prospective registrar. E.g., the certification bodies in North America will be certified through an organization such as the ANSI/ASQ ANAB. Accreditation boards are formed for every major nation to review certification organizations to ensure they meet the standards.
The target demographic should always be held in mind. For example, if a medical product supplier needs to sell in North America, he would seek the authorization of a registrar approved to conform with the country or customer standards from a North American accreditation body.
IQC The ISO Pros can help you implement, train on, consult, and have your company and/or organization certified on the following ISO Certifications: